Applications
Medical
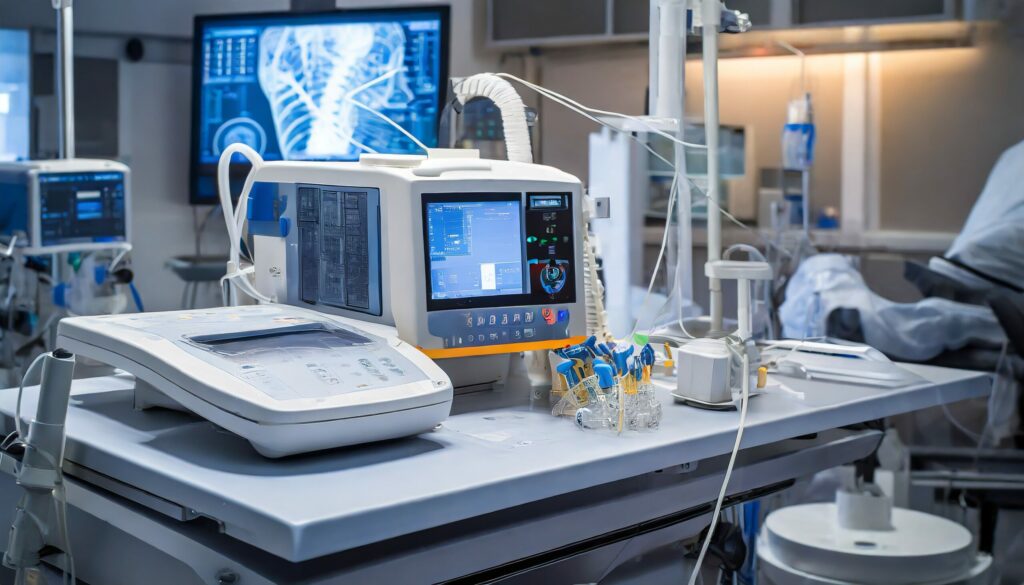
As experts in the medical device field, we recognize the risks associated with the development of electronic medical devices. Therefore, Omnitek adheres to the highest standards required by the healthcare industry and provide our clients with the necessary documentation and resources for them to succeed. We have wide experience with innovative medical applications, where solutions require high-reliability results, risk assessments, documentation, and FDA approvals. Whether a project requires turnkey development, partial development, or a proof-of-concept, Omnitek provides the electronics to keep patients alive by design.
Commercial
Omnitek understands that time-to-market and product quality are critical to our clients. Therefore, our engineers collaborate with parts vendors, contract manufacturers, and other development partners, in order to guarantee that these goals will be achieved prior to the launch of the development process. Beginning with the consumer in mind, Omnitek works to connect the electronics with the end-user, in order to create a well-designed, reliable product. Effective, innovative solutions are used to enhance the success of a client’s product for the competitive global marketplace.

Industrial Control
Omnitek offers Embedded Firmware and Software design solutions with expertise in requirements analysis, system architectures, code development, and integration testing, spanning various markets and applications such as communications, user interfaces, data acquisition, and device control.

Mobile Applications
Omnitek develops mobile applications that facilitate communication between electronic devices. These customized applications can utilize any of the popular platforms–iOS, Linux, and Android. Let us know your needs.

